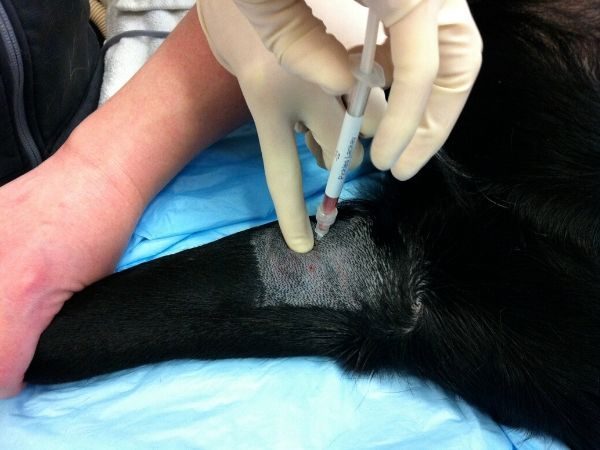
Overview of Joint Injections
Osteoarthritis is currently an incurable and irreversible condition, but there are a lot of tools that can be effective at managing the progression and symptoms.
Intra-articular (IA) therapy, i.e. joint injections, have traditionally been recommended if non-invasive therapies such as medication, weight loss, supplements, and rehabilitation modalities have been not sufficient to reduce pain associated with arthritis. However, there are emerging arguments for using some IA products early in the course of OA rather than end stage, as some products may be more efficacious in mild to moderate OA. At this time there are no studies comparing all of the available IA products in terms of efficacy or safety or providing clear guidance on timing of injections. Nonetheless, IA therapies are an important aspect of an OA plan and are particularly useful for managing elbow OA.
How to administer joint injections
There are currently several categories of IA therapies and new classes of products are being developed. No matter what product is injected, animals must be sedated for the procedure in order to ensure strict sterility for injection and precise injection into the joint.
There is a learning curve for performing joint injections, and some joints are more easily accessed than others. When starting to perform joint injections, it is highly recommended that the animal be deeply sedated in order to minimize movement and discomfort to the patient.
Radiographs and ultrasound can be used to guide needle insertion, though once the skill has been mastered, these diagnostics should not be needed. Once you become proficient in joint injections, there will be many animals that can be treated under light sedation (gabapentin/trazodone/butorphanol) and a local block. The frequency of repeating injections will likely vary with the product used, and even then, there are no clear guidelines in animals for the frequency of treatment.
Intra-articular Injectable Products
Platelet Rich Plasma (PRP): Platelets contain a large number of growth factors and proteins that modulate inflammation, healing, and the immune system. We know that there is a component of osteoarthritis that involves increased activity of certain immune cells and increased production of IL-1 and TNF-alpha.
PRP therapy involves collecting the dog’s own blood and isolating the platelets using a commercially available syringe and centrifuge. Each commercial product has a different collection and processing method, but they can all be done in the hospital (do not need to send out to a lab). The processing generally takes about 20 minutes.
- There are currently 3 products available in the US that have research from clinical trials that show positive results in dogs with OA: Autologous Conditioned Plasma (ACP, Arthrex), Protein Solution (APS/Pro-Stride, Zoetis), and PALL V-Pet (Vet Stem) (Franklin S 2013, Fahie MA, 2013, Wanstrath AW 2016).
- There are several other PRP products on the market that do not yet have clinical evidence to support their use in dogs with OA. There are no studies comparing the different products, and each product contains a different concentration of platelets, white blood cells, and red blood cells.
- There is currently a debate within human and veterinary sports medicine as to the optimal concentration of platelets and WBC (leukocyte-poor vs. leukocyte-rich). Until clear evidence supports one product over others, we recommend choosing one of the 3 products listed above that have undergone clinical research in dogs.
- Studies suggest that clinical effects are seen between 1-3 months after injection. Studies with the APS and V-Pet systems showed significant improvement in weight-bearing as measured by force plate (in fact, the degree of improvement was more than is expected with NSAIDs!). Client-assessed outcomes, using the CBPI, also showed significant improvement. It is currently unknown how long the positive effects last, but repeat injections are likely needed every 6-12 months.
Stem Cell Therapy: Stem cell therapy for veterinary patients utilizes adult, or mesenchymal, stem cells. Currently, these cells are derived from an animal’s own fat or bone marrow (autogenous) and are capable of limited differentiation (as opposed to embryonic stem cells).
Allogeneic stem cell therapy, using a donor dog’s cells in an “off-the-shelf” product is currently being investigated. The currently available stem cell therapies are typically a combination of mesenchymal stem cells and other cells such as endothelial progenitor cells, M2 macrophages, and T-regulatory cells.
Some therapies will culture mesenchymal stem cells and deliver these cells alone. It is unknown whether the “soup” of cells is more or less effective than mesenchymal stem cells alone. One important concept is that to date, no type of stem cell therapy has been proven to be “regenerative,” so the phrase “regenerative medicine” is inappropriate and misleading to use.
Cell-based therapy is very likely the way of the future for the management of not only OA but many disease conditions. This is a very active area of research. There are several studies that have documented positive response to mesenchymal stromal cell therapy (ie, the “soup”) in dogs with OA. Stay tuned for updates as more evidence becomes available!
Hyaluronic Acid (HA): HA is a normal component of cartilage and joint fluid that provides lubrication. In arthritic joints, the HA molecule is smaller than normal leading to a less viscous fluid. Intra-articular injection of synthetic HA is believed to alter the metabolism of naturally occurring HA and decrease symptoms of OA.
There remains debate in human and veterinary sports medicine as to the efficacy of HA. This debate stems from a wide variety of study designs and products that have been tested. HA products primarily differ in their molecular weight and are categorized as Low Molecular Weight (LMW, 500-730kDA) or High Molecular Weight (HMW, ~6,000kDA).
HMW more closely resembles normal HA in non-arthritic joints (5,000-10,000kDA). A study was conducted in dogs that had experimentally induced OA and compared HMW and LMW products as well as one injection to 3 injections. (Pashuck TD, 2016) Results found that both products and both treatment protocols were significantly better than saline control; however, the HMW was better than LMW. The maximal benefit in pain, lameness, function, and range of motion was seen 4-8 weeks after injection and began to subside at 6 months.
Steroids (triamcinolone): Steroid injections are a common treatment of arthritis in people and animals. However, we know that repeated steroid injections can be detrimental to the cartilage, so we typically recommend reserving steroid injections as a last resort for “end-stage” disease when there is little to no cartilage remaining. Steroids can be effective in decreasing inflammation and pain in the joint. Steroids are typically combined with HA when administered. One study showed equivalent results of HA plus steroids vs. ACP (Arthrex brand of PRP). Improvements were seen as soon as 1 week after injection and lasted at least 12 weeks (Franklin SP 2013).
References
Franklin SP, Cook JL. Prospective trial of autologous conditioned plasm versus hyaluronan plus corticosteroid for elbow osteoarthritis in dogs. Can Vet J 2013;54:881-884.
Fahie MA, Ortolano GA, Guercio V, et al. A randomized controlled trial of the efficacy of autologous platelet therapy for the treatment of osteoarthritis in dogs. J Am Vet Med Assoc 2013;243:1291-1297.
Wanstrath AW, Hettlich BF, Su L, et al. Evaluation of a single intra-articular injection of autologous protein solution for treatment of osteoarthritis in a canine population. Vet Surg 2016;45:764-774.
Arnhold S, Wenisch S. Adipose tissue-derived mesenchymal stem cells for musculoskeletal repair in veterinary medicine. Am J Stem Cells 2015;4:1-12.
Frisbie DD, Kisiday JD, Kawcak CE, et al. Evaluation of adipose-derived stromal vascular fraction or bone marrow-derived mesenchymal stem cells for treatment of osteoarthritis. J Orthop Res 2009.
Black LL, Gaynor J, Gahring D, et al. Effect of adipose-derived mesynchymal stem and regenerative cells on lameness in dogs with chronic osteoarthritis of the coxofemoral joints: A randomized, double-blinded, multicenter, controlled trial. Vet Therapeutics 2007.
Black LL, Gaynor J, Adams C, et al. Effects of intraarticular injection of autologous adipose-derived mesenchymal stem and regenerative cells on clinical signs of chronic osteoarthritis of the elbow joint in dogs. Vet Therapeutics 2008.
Harman R, Carlson K, Gaynor J, et al. A prospective, randomized masked, and placebo-controlled efficacy study of intraarticular allogeneic adipose stem cells for the treatment of osteoarthritis in dogs. Frontiers Vet Sci 2016.
Upchurch DA, Renberg WC, Roush JK, et al. Effects of administration of adipose-derived stromal vascular fraction and platelet-rich plasma to dogs with osteoarthritis of the hip joints. Am J Vet Res 2016;77:940-951.
Kuroki K, Cook JL, Kreeger JM. Mechanisms of action and potential uses of hyaluronan in dogs with osteoarthritis. J Am Vet Med Assoc 2002;221:944-950.
Canapp SO, Cross AR, Brown MP, et al. Examination of synovial fluid and serum following intravenous injections of hyaluronan for the treatment of osteoarthritis in dogs. Vet Comp Orthop Traumatol 2005;18:19-174.
Smith GN, Mickler EA, Myers SL, Brandt KD. Effect of intraarticular hyaluronan injections on synovial fluid hyaluronan in the early stage of canine post-traumatic osteoarthritis. J Rheumatol 2001;28:1341-1346.
Pashuck TD, Kuroki K, Cook CR, et al. Hyaluronic acid versus saline intra-articular injections for amelioration of chronic knee osteoarthritis: A canine model. J Orthop Res 2016; 34:1772-1779.
Carapebe GOL, Cavaleti P, Nicacio GM, et al. Intra-articular hyaluronic acid compared to traditional conservative treatment in dogs with osteoarthritis associated with hip dysplasia. Evidence-Based Comp Alt Med 2016.