Guest article by:
Patrice M. Mich, DVM, MS, DABVP, DACVAA, DACVSMR, CCRT
Dr. Patrice Mich is a small animal pain management and mobility specialist. Her career focuses on regaining and maintaining functional independence and performance for canine athletes of all ages and abilities. In Colorado, where Dr. Mich calls home, almost all dogs are athletes, amateurs, and professionals. She is a graduate of Colorado State University School of Veterinary Medicine. During 14 years as a generalist, she completed advanced training in veterinary acupuncture and achieved diplomate status in the American Board of Veterinary Practitioners, canine/feline practice.
As veterinarians, we see many cases of osteoarthritis every day. So much so that we can be taken by surprise when a less common problem like immune-mediated polyarthropathy (IMPA) presents. In this brief review, I cover clinical presentation, diagnosis, and treatment. Categories of polyarthropathy will be reviewed as well. An easy way to group all polyarthropathies broadly is
- Infectious causes (bacterial, mycoplasma, tick-borne disease, fungal, and viral organisms);
- Noninfectious, non-erosive immune-mediated causes
- Noninfectious erosive causes. Another method of categorizing IMPA specifically is listed below.
Types of IMPA include non-erosive and erosive (sometimes called “rheumatoid”) types. The former include type I or “idiopathic”; type II associated with infectious disease (e.g. fungal, viral); type III associated with gastrointestinal disease, and type IV associated with conditions such as neoplasia, vaccination reactions, and systemic lupus. Erosive IMPA causes bone and soft tissue destruction and is less common; the underlying mechanism(s) of this condition has not been determined.
 Clinical presentation of polyarthropathy has some distinct features compared to multiple joint osteoarthritis. Both involve inflammatory processes, but the clinical pathology and clinical signs are quite different. Nonspecific systemic signs including malaise, weight loss, hyporexia, and reluctance to move are notable. Additionally, some dogs present with a stiff or “walking on eggshells” gait in which stride length is markedly shortened for affected limbs. Shifting leg lameness also occurs. Although some dogs (and cats) present with joint effusion, others present with systemic illness and no obvious localizable pain or swelling. IMPA is one of the most common causes of fever of unknown origin. It is also mistaken for cervical or spinal pain due to disc disease or meningitis because facet joints can be affected as well. Importantly, by definition, IMPA involves more than 1 joint, but early in the course, clinical signs may be limited to one joint.
Clinical presentation of polyarthropathy has some distinct features compared to multiple joint osteoarthritis. Both involve inflammatory processes, but the clinical pathology and clinical signs are quite different. Nonspecific systemic signs including malaise, weight loss, hyporexia, and reluctance to move are notable. Additionally, some dogs present with a stiff or “walking on eggshells” gait in which stride length is markedly shortened for affected limbs. Shifting leg lameness also occurs. Although some dogs (and cats) present with joint effusion, others present with systemic illness and no obvious localizable pain or swelling. IMPA is one of the most common causes of fever of unknown origin. It is also mistaken for cervical or spinal pain due to disc disease or meningitis because facet joints can be affected as well. Importantly, by definition, IMPA involves more than 1 joint, but early in the course, clinical signs may be limited to one joint.
The most commonly affected joints are carpus, tarsus, stifle, and elbow. However, any joint can be affected. Erosive IMPA causes bone destruction and in advanced cases, joint collapse of digits, carpi, and tarsi are not uncommon. This is due to subsequent damage to supporting soft tissues. Non-erosive IMPA presents with effusion or no effusion, joint pain or no pain, and no joint instability or bone destruction.
Diagnosis of IMPA
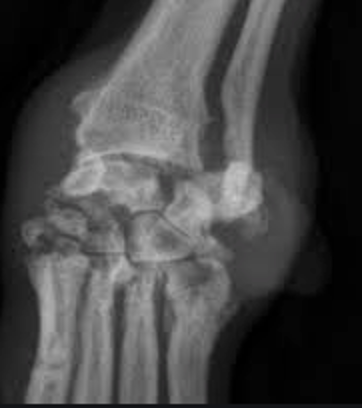
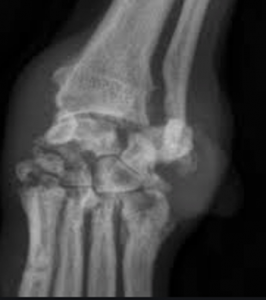
Diagnosis of IMPA begins with a level of suspicion based on the above clinical signs. Certainly, joint collapse as is seen with erosive forms is telling as are supportive radiographs revealing bone lysis (so-called “punched out” lesions”). In order to confirm and categorize non-erosive IMPA, a complete work up guided by clinical presentation including the steps listed in box 2 is ideal.
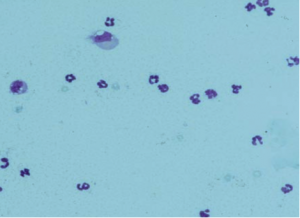
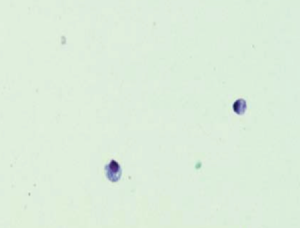
 This extensive workup can be helpful in excluding many etiologies. That said, from a practical perspective, cytologic evaluation of joint fluid can confirm IMPA allowing for empirical treatment when a full workup is not possible; arthrocentesis of 3 joints is recommended. Normal joint fluid should have less than 3000 WBC/ ml and rare RBCs. Mononuclear cells make up 90% of the WBCs. Inflamed joints will have higher WBC counts and greater than 10% neutrophils. An easy method of screening in clinic is to count the number of cells per high-powered field and multiply by 1000. Septic joints will have large numbers of degenerative neutrophils and you will often see bacteria extra or intracellularly. When seeking etiology pooled culture is sufficient and the use of broth enriched media ( e.g. blood culture bottles) increases success in culturing.
This extensive workup can be helpful in excluding many etiologies. That said, from a practical perspective, cytologic evaluation of joint fluid can confirm IMPA allowing for empirical treatment when a full workup is not possible; arthrocentesis of 3 joints is recommended. Normal joint fluid should have less than 3000 WBC/ ml and rare RBCs. Mononuclear cells make up 90% of the WBCs. Inflamed joints will have higher WBC counts and greater than 10% neutrophils. An easy method of screening in clinic is to count the number of cells per high-powered field and multiply by 1000. Septic joints will have large numbers of degenerative neutrophils and you will often see bacteria extra or intracellularly. When seeking etiology pooled culture is sufficient and the use of broth enriched media ( e.g. blood culture bottles) increases success in culturing.
Radiographs can reveal erosive lesions sometimes called “punched out” regions as well as significant joint collapse in advanced cases.
Complete workup is intended to identify definitive etiology and if none is found idiopathic IMPA is the presumptive diagnosis. Importantly septic arthritis must be ruled out before immunosuppressive therapy is instituted.
 Treatment of IMPA is based on the underlying cause if identified. Additional therapy must address pain. The cornerstone of idiopathic IMPA therapy is immunosuppressive medication and, in general, prognosis is good although relapse can occur. Glucocorticoids (once bacterial etiology is ruled out) are typically first-line therapy followed by the addition of or transition to other immunosuppressive agents ( Box 3). For patients with mild and transient polyarthritis, NSAIDs may be sufficient, but should not be given concurrently with glucocorticoids.
Treatment of IMPA is based on the underlying cause if identified. Additional therapy must address pain. The cornerstone of idiopathic IMPA therapy is immunosuppressive medication and, in general, prognosis is good although relapse can occur. Glucocorticoids (once bacterial etiology is ruled out) are typically first-line therapy followed by the addition of or transition to other immunosuppressive agents ( Box 3). For patients with mild and transient polyarthritis, NSAIDs may be sufficient, but should not be given concurrently with glucocorticoids.
Uncomplicated cases and those without erosive disease may be tapered down or off medications slowly. The decision to taper is ideally based on repeat cytologic evaluation, but if not possible signification decrease in or resolution of clinical signs for at least 4 weeks has been used by this author to guide initiation of slow tapering. Dosage change every 4 weeks is a good rule of thumb. If clinical signs recur increase in dose or additional medications may be necessary. Remission can be established in some cases. Notably, in recent years use of leflunomide as monotherapy has allowed for decrease in adverse effects resulting from immunosuppressive doses of glucocorticoids. Additionally, the use of non-glucocorticoid immunosuppressive agents provides the opportunity to utilize NSAIDs for pain management. If a patient is non-responsive to one agent switching to an alternative may provide satisfactory results. Note that in general erosive IMPA is more difficult to treat; refractory cases are not uncommon and long-term prognosis is poor.
Pain management for IMPA patients must be provided. As noted above NSAIDs are useful under some circumstances. Other therapies include gabapentin, acetaminophen, amantadine, manual therapy, and LASER.
Lastly, empirical therapy with doxycycline is commonly provided. This is because L-form bacteria and mycoplasma are difficult to identify. Doxycycline is also used to treat infectious agents such as Ehrlichia, Lyme’s disease, anaplasmosis, and Rocky Mountain spotted fever.
Polyarthropathy in cats occurs much less frequently than degenerative joint disease (osteoarthritis) just as it does in dogs. A significant difference is that infectious causes are more frequent in cats; idiopathic IMPA is considered rare. Clinical signs of feline inflammatory joint disease are similar including systemic signs such as lethargy, pyrexia, and anorexia. Lameness is a common clinical finding and joint effusion occurs in many cases, but not all. Diagnostic evaluation is similar to that of dogs with the addition of FeLV and FIV testing. Treatment of infectious agents is ideally based on definitive identification of the organism. Broad-spectrum antibiotics are prescribed for more common organisms such as Staphylococcus, Streptococcus, and Pasteurella spp. Doxycycline is used for specific or empirical treatment for less common infectious agents described above. A rare and aggressive syndrome of rheumatoid-like arthritis has been described in cats. Prognosis is poor. A second rare condition seen most often in young males is feline periosteal proliferative polyarthritis. Response to glucocorticoids can decrease severity of signs although slow progression is expected.
References
1. Jacques D, Cauzinille L, Bouvy B, et al. A retrospective study of 40 dogs with polyarthritis. Vet Surg 2002;31(5):428–34.
2. Johnson KC et a. Canine Immune-Mediated Polyarthritis Part 1 and 2. JAAHA 2012; 48(2): 71-82.
3. Bennet D. Immune-mediated and infectious arthritis. IN: Ettinger SJS, Feldman EC eds. Textbook of veterinary internal medicine, vo2 , 6th edition. St Louis (MO): Elsevier Sanders; 2005: 1958-64.
4. Taylor SM. Arthrocentesis. In: Taylor SM, ed. Small animal clinical techniques. St Louis (MO): Elsevier Sanders; 2009: 189-201.
5. Shaughnessy ML et al. Clinical features and pathological joint changes in dogs with erosive immune-mediated polyarthritis. (2004-2012). JAVMA 2016; 249: 1156-1164.
6. Colopy, SA et al. Efficacy of leflunomide for treatment of immune mediated polyarthritis in dgos: 14 case (2006-2008). JAVMA 2010; 236(3): 312-218.
7. Gregory CR. Immunosuppressive agents. In: Bonagura JD, Twedt DC, eds. Kirk’s current veterinary therapy XIV. St Louis (MO): Elsevier Sanders; 2009: 254-259.
8. Inflammatory Joint Disease in Cats: diagnostic approach to treatment. JFMS 2014; 16: 547-562.